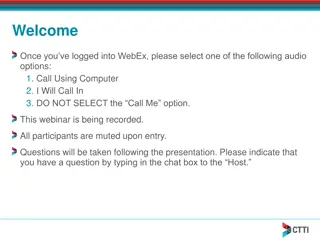
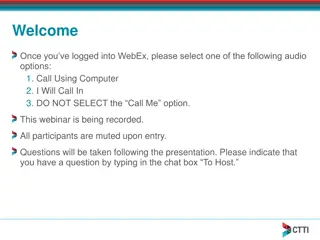
Staff and Trials Overview at KVK Champhai, Mizoram
KVK Champhai, Mizoram, has a diverse staff including scientists and support staff. They conduct on-farm trials in disciplines like agronomy, horticulture, and plant protection. Trials aim to refine and assess technologies for crops like maize, beans, soybean, and more under various conditions. Speci
2 views • 49 slides
Infrastructure Trials for Technology Coordinators - Overview and Logistics
Explore the essential aspects of conducting infrastructure trials for technology coordinators in educational settings. The session covers topics such as determining the need for trials, tasks for technology staff, resources and support available, and live sandbox time for practical application. Pres
2 views • 58 slides
Overview of UW Clinical Trial Office Budget Review
UW Clinical Trial Office conducts budget reviews to ensure compliance and financial accountability in clinical trials. The office collaborates with various departments to manage billing compliance, financial risks, and institutional policies. The primary focus is on avoiding patient billing errors,
0 views • 17 slides
Market Trials Planning Overview
Market Trials Planning Overview provides a detailed timeline and procedures for the upcoming trials in the RTC+B program, including activities such as QSE Attestation, Market Submission Testing Objectives, and Transition to Go-Live. The document outlines potential dates, testing requirements, and ke
0 views • 12 slides
Ethical Issues in Clinical Pharmacy Research by Dr. Haider Raheem Mohammad
Research ethics play a crucial role in clinical trials and therapeutic research in the field of pharmacy. From discovery to validation, all medicines undergo rigorous evaluation processes to ensure safety, efficacy, and freedom from adverse effects. Clinical trials in both animals and humans are ess
0 views • 20 slides
Understanding Participant Rights, Responsibilities, and the Role of Patient Organisations in Clinical Trials
Participants in clinical trials have specific rights that are protected by law, including the right to informed consent and the right to be informed of any new benefits, risks, or side effects discovered during a study. They also have responsibilities such as adhering to the prescribed medication sc
0 views • 8 slides
NIMH Clinical Research Education and Monitoring Program Overview
NIMH's Clinical Monitoring and Clinical Research Education, Support, and Training Program (CREST) aims to ensure the proper conduct, recording, and reporting of clinical trials. This program includes clinical monitoring plans, guidelines for site monitoring activities, and independent clinical monit
1 views • 29 slides
Understanding Ethics in Clinical Trials: A Comprehensive Overview
Explore the historical context, important ethical guidelines, and the ethical framework with 7 principles in the field of clinical trials. Learn about key trials, ethical considerations, and guidelines governing human subject research in clinical medicine. Delve into the critical aspects such as inf
1 views • 15 slides
Unified Stata Package for Sample Size Calculations in Clinical Trials
A comprehensive overview of artbin, a Stata package for determining sample sizes in trials with binary outcomes. This tool is essential for designing effective clinical trials across various disciplines and has recently undergone significant enhancements. Unlike other software, artbin offers a wide
1 views • 23 slides
Understanding Non-Clinical Development in Therapeutic Innovation
The European Patients Academy on Therapeutic Innovation focuses on the non-clinical development phase of medicine, delving into efficacy assessment, safety evaluation, and manufacturing process considerations. Non-clinical studies are essential for decision-making in clinical trials, marketing appli
1 views • 26 slides
Ensuring Protocol Compliance and Corrective Action Plans in Clinical Trials
This content discusses the importance of creating Corrective Action and Preventive Action (CAPA) plans for protocol deviations in clinical trials. It covers the components of a CAPA, best practices for creating CAPAs for different deviation types, and regulatory compliance requirements according to
0 views • 33 slides
Understanding Essential Documents in Clinical Trials
This presentation by Derita Bran, BSN, RN, CCRC, focuses on the documentation required for quality clinical trials, emphasizing Essential Documents outlined in the ICH GCP E6 (R2) guidance. It covers the purpose, definition, and importance of these documents in ensuring subject rights protection, da
4 views • 35 slides
Understanding Investigational Products in Clinical Trials
Investigational products play a crucial role in clinical trials, encompassing drugs, devices, biologics, and more. The FDA defines investigational new drugs as substances seeking approval, even if previously in use, with potential changes. Similarly, investigational devices are those under investiga
0 views • 43 slides
European Patients Academy on Therapeutic Innovation: Role of Statistics in Clinical Trials
European Patients Academy on Therapeutic Innovation emphasizes the significant role of statistics in clinical trials. Clinical research aims to determine treatment efficacy and safety, while statistical methods aid in designing, analyzing, and reporting trials to minimize biases and errors. Understa
1 views • 5 slides
BMC Clinical Trials Office Goals and Initiatives
The BMC Clinical Trials Office (CTO) is dedicated to supporting clinical and human research at Boston Medical Center by providing leadership and expertise in research, finance, and administration. The CTO reviews, negotiates, and approves protocols, supports grants and contracts, ensures accurate cl
0 views • 17 slides
Understanding Clinical Trials: Types and Designs
Clinical trials are essential research studies that evaluate new tests and treatments to improve human health outcomes. They involve various phases, designs, and purposes, such as treatment trials, prevention trials, and observational studies. Different types of clinical trial designs include experi
7 views • 18 slides
Veterinary Medicinal Products Clinical Trials in Ireland
This presentation covers the process and regulations surrounding clinical trials and testing of veterinary medicinal products in Ireland. Topics include the application process, HPRA evaluation, fees, scope of legislation, and the purpose of clinical trials. Feedback from a 2016 HPRA survey on clini
3 views • 17 slides
Understanding Compensation and Reimbursement in Clinical Trials
Compensation in clinical trials can involve monetary benefits for participation or services for harm suffered. It is more common in Phase I trials with healthy volunteers. Reimbursement covers expenses like travel, accommodation, and loss of income for participants. Legislation in Europe varies on c
1 views • 16 slides
Market Trials Overview for SPP Members
This document outlines the market trials conducted by Southwest Power Pool (SPP) to help its members collaborate in ensuring a reliable power supply. It covers weekly status meetings, agenda details, test schedules, key points, and future steps. The purpose of blast calls and the market trials calen
1 views • 26 slides
Brain Oxygen Optimization in Severe Traumatic Brain Injury (BOOST3) Trials Overview
Overview of the BOOST3 trials focusing on brain oxygen optimization in severe traumatic brain injury patients. The trials involve multiple PIs, training sessions, and hands-on ancillary studies. Various design principles and organizational values are emphasized, with grant awards distributed to diff
0 views • 13 slides
Enhancing Patient Engagement in Clinical Trials
This presentation highlights the importance of patient engagement in clinical trials, showcasing the value it brings to the drug development process. It covers various aspects such as quantifying impact, identifying opportunities, and the role of patient groups. The Clinical Trials Transformation In
0 views • 37 slides
Insights into the Salem Witch Trials
Discover the dark history of the Salem Witch Trials, where people were accused of witchcraft based on superstitions and fear. Learn about the beliefs, trials, possible explanations, options for the accused, and the chaos that ensued in Salem. The trials lasted from January 1692 to May 1693, revealin
0 views • 11 slides
Challenges and Recommendations for DHT-derived Endpoints in Clinical Trials
This study by CTTI explores barriers and solutions for adopting digitally derived endpoints in clinical trials. Through in-depth interviews with industry sponsors, the research identifies gaps, barriers, and recommendations for using DHT-derived novel endpoints as key endpoints in pivotal clinical t
0 views • 16 slides
Clinical Research Guidelines and Regulations Overview
Clinical research encompasses various guidelines and regulations to ensure the protection of human subjects and the credibility of study results. Key aspects include Good Clinical Practice (GCP) standards, Title 45 of the Code of Federal Regulations (CFR) Part 46, and additional CFR sections for cli
0 views • 46 slides
Insights into the Salem Witch Trials
Explore the historical beliefs around witchcraft, the occurrences of the Salem witch trials, possible explanations for the trials, the options accused witches had, how events quickly spiraled out of control in Salem, and the duration of the trials from January 1692 to May 1693.
0 views • 10 slides
Enhancing Clinical Trials through ICH E6 Renovation
Explore the Clinical Trials Transformation Initiative's efforts in updating ICH E6 guidelines to improve global clinical trials. Learn about CTTI's findings, insights from stakeholders, and discussions on the necessary updates for ICH E6. Engage with industry experts and regulatory authorities to un
0 views • 65 slides
Exploration of Endpoints in Clinical Trials for Alcohol Use Disorder
This presentation discusses the exploration of endpoints in pivotal clinical trials to treat Alcohol Use Disorder (AUD), focusing on Primary Alcohol Drinking Endpoints like PSNHDD. Evidence supporting PSNHDD as a primary endpoint includes clinical benefit data from alcohol clinical trials and epidem
0 views • 54 slides
Embracing Trials: A Biblical Perspective on Enduring Hardships
Understanding the role of trials in a Christian's life through the teachings of Peter. Be prepared to face trials, rejoice in Christ's sufferings, and never be ashamed of your faith amidst hardships. Others face trials too, so be encouraged and remain faithful.
0 views • 12 slides
Key Aspects of Partnering with a Clinical Trial Unit for Successful Trials
Understanding the role and benefits of partnering with a Clinical Trial Unit (CTU) is essential for potential Chief Investigators. A CTU handles various aspects of trial coordination, from developing trial materials to overseeing regulatory permissions and data management. UK Clinical Trials Units (
0 views • 48 slides
Statistical Issues in Clinical Trials: Insights from 13th Annual Conference
The 13th annual conference on Statistical Issues in Clinical Trials covered topics such as penalties for extra variation and limited degrees of freedom, the Diet-Heart Hypothesis, controlled trials, unit of randomization, and causal inference. Speakers highlighted the importance of addressing cluste
0 views • 10 slides
Understanding Craving as an Outcome Measure in OUD Trials
Craving plays a vital role in substance use disorder (SUD) diagnosis, with intense desires or urges triggered by environmental cues. The focus is on identifying and validating the best craving measure for OUD clinical trials. Near-term goals include qualifying craving as a Clinical Outcome Assessmen
0 views • 12 slides
Challenges in Missing Data Handling in Clinical Trials
Long-standing approaches for handling missing data in clinical trials are being questioned, leading to discussions on the appropriateness and implications of methods like MMRM. Regulators emphasize the importance of selecting methods based on the trial's context rather than the method's properties.
0 views • 34 slides
Introduction to Clinical Trials and Important Terms
This content provides an overview of clinical trials, including the goals, phases, study funding, and important terms such as placebo. Clinical trials aim to study the impacts of health and disease, evaluate treatments, and ensure safety and effectiveness. The phases of clinical trials are explained
0 views • 27 slides
Non-Malignant Haematology Clinical Trials at Hammersmith Hospital: Latest Research Updates
Explore the latest updates from the Non-Malignant Haematology Clinical Trials Unit at Hammersmith Hospital, including studies on Red Cell Disorders and the REDRESS trial for severe sickle cell disease. Learn about ongoing trials, inclusion criteria, and contact information for participation.
0 views • 11 slides
Therapeutic Trials in HIV/HCV Coinfection: ION-4, Ally-2, and Viroteam 2015
In the study of therapeutic trials in HIV/HCV coinfection between 2014-2015, notable trials included ION-4 with LDV/SOF treatment for 12 weeks in HCV/HIV co-infection patients, showing high SVR rates in both treatment-naive and experienced patients. Another trial, Ally-2, involved Daclatasvir and So
0 views • 15 slides
Understanding Protocol Deviations in Clinical Trials
Protocol deviations are expected occurrences in clinical trials that may arise from site or participant actions. It is crucial for research teams to identify, report, and learn from these deviations to ensure the integrity and safety of the study. Mechanisms for identifying and reporting deviations,
0 views • 16 slides
Understanding Clinical Trial Endpoints and Regulatory Basis
Eugene J. Sullivan, MD, explains the importance of establishing efficacy in clinical trials based on substantial evidence from well-controlled trials. The discussion includes primary endpoints for Phase 3 trials, legal and regulatory requirements, and the significance of clinically meaningful effect
0 views • 21 slides
NHMRC's New Grant Program: Advancing Clinical Trials and Research Funding
The NHMRC's latest grant program aims to enhance research in healthcare by focusing on clinical trials funding across four key streams: Investigator Grants, Synergy Grants, Ideas Grants, and Strategic and Leveraging Grants. The redistribution of funding will see a significant increase in support for
0 views • 12 slides
Clinical Trials Market Forecast to Grow to $102.20 Billion by 2031, Fueled by Ad
Explore $102.20 billion Clinical Trials Market: Get exclusive insights on key market trends, segments, geographical analysis, & competitive analysis!
1 views • 9 slides
Understanding Clinical Trials: Phases, Types, and Definitions
Clinical trials play a crucial role in advancing medical research and treatment options. This comprehensive guide covers the basics of clinical trials, including their definition, phases, types, and key definitions like IND, IDE, NDA, and more. Discover how different phases of trials work, the vario
0 views • 16 slides