Preparation of Aspirin: Overview and Synthesis Methods
The preparation of aspirin involves the synthesis of 2-acetyl salicylic acid, known for its therapeutic uses as an analgesic, antipyretic, and anti-inflammatory agent. This process includes the detailed characteristics, stability, mechanism, and synthesis methods of aspirin. Important considerations, such as stability in dry air, degradation with moisture, and decomposition of aspirin tablets, impact its pharmaceutical quality. The synthesis methods illustrate the chemical reactions for producing aspirin, providing insights into the protonation steps involved.
Download Presentation

Please find below an Image/Link to download the presentation.
The content on the website is provided AS IS for your information and personal use only. It may not be sold, licensed, or shared on other websites without obtaining consent from the author.If you encounter any issues during the download, it is possible that the publisher has removed the file from their server.
You are allowed to download the files provided on this website for personal or commercial use, subject to the condition that they are used lawfully. All files are the property of their respective owners.
The content on the website is provided AS IS for your information and personal use only. It may not be sold, licensed, or shared on other websites without obtaining consent from the author.
E N D
Presentation Transcript
Preparation of aspirin By Assistant lecturer : Dina Saleem
Therapeutic uses Aspirin act as an analgesic, antipyretic, Anti-inflammatory also inhibit platelet aggregation & prolongs bleeding time, because of its effect on G.I.T it is contraindicated in peptic ulcer, in this case we use paracetamol . Aspirin is not given to children because is may cause raye s syndrome.
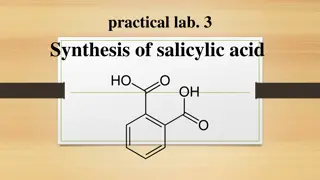
Preparation of aspirin It is 2-acetyl salicylic acid or 2-acetoxy benzoic acid. Molecular formula is C9H8O4, M wt is 180.15. Characteristics: It is colorless crystals or white crystalline powder , odorless with slightly acid taste. Solubility: It is slightly soluble in water (1:300) at 25 , (1:100) at 37 more soluble in organic solvent, e.g. ether , ethanol, chloroform, .. etc.
Stability: In the form of crystals or powder, ASA is stable in dry air , but in contact with moisture, it degrades by hydrolysis to both SA and acetic acid (HAc), through base catalysis of the attack of water molecule by carboxylate anion. Mechanism:
Extensive decomposition of aspirin tablets is easily recognized by the odor of acetic acid , partially degraded aspirin tablets are sometimes seen with needles SA on their surface, a result of sublimation of SA. The melting point of ASA is not a reliable indicator of its purity because it partly decomposes on heating (137-143 ).
Synthesis of aspirin: Method 1:
Mechanism of reaction: 1- Protonation of the oxygen of carbonyl group:
Procedure :- 1-Place 3 gm of salicylic acid in 100 ml E.f. and add with constant stirring 6 ml of acetic anhydride followed by 1 ml of conc. H2SO 2- Stir the mixture gently observing the rise in temp. to 70-80 oC. While the salicylic acid dissolves, after 15 minutes the solution cools by it self and a solid mass of aspirin forms 3-Pour 35 ml of ice-cold water over the contents of the flask to hydrolyze excess acetic anhydride and to complete the ppt of aspirin 4- Collect the crude aspirin using a Buchner funnel and wash with ice-cold water, air-dry the product and calculate the yield 5- Perform FeCl3 test on produced aspirin
Identification test of aspirin :- Ferric chloride test; The presence of phenolic group in a compound is indicated by the formation of violet iron complex when treated with ferric chloride solution Aspirin ------------ give ve result with FeCl3 due to absence of phenolic group Salicylic acid-------- + ve result
Notes: Method 1 is used mostly because acetic anhydride is cheap, readily available, easily handled and doesn't form corrosive HCl gas. The reaction of acetylation is in the following order: Acetyl chloride > acetic anhydride > acetic acid On the other hand, acetylation of SA using acetyl chloride is very vigorous and will produce HCl gas which is corrosive to the respiratory tract. In this method, pyridine base is used because the unshared pair of electrons on the N will react with HCl and form pyridine chloride. On the other hand , pyridine base is teratogenic agent and air pollutant; therefore, we avoid this method of preparation.