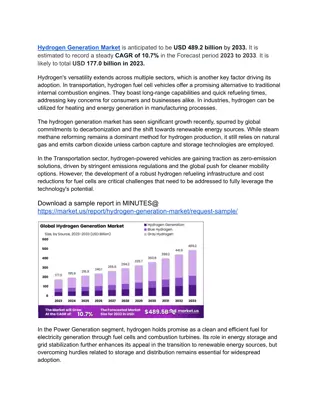
Exploring Hydrogen and Its Compounds: Properties and Isotopes
Hydrogen, the most abundant element in the universe, has unique properties that make it resemble both alkali metals and halogens. It exists mainly in combined states, except in volcanic gases, and its isotopes exhibit distinct chemical and physical characteristics. The ionization energy of the H-H bond is high, indicating low tendency to form H+ ions. Hydrogen's isotopes share same chemical properties but differ significantly in other aspects like mass numbers and reactivity levels.
Download Presentation

Please find below an Image/Link to download the presentation.
The content on the website is provided AS IS for your information and personal use only. It may not be sold, licensed, or shared on other websites without obtaining consent from the author. Download presentation by click this link. If you encounter any issues during the download, it is possible that the publisher has removed the file from their server.
E N D
Presentation Transcript
HYDROGEN AND IT S COMPOUNDS Dr. Nouf H. Alotaibi
HYDROGEN AND ITS COMPOUNDS Introduction: Hydrogen is the most abundant element in the universe. It is the 9th most abundant element in the earth's crust. It occurs only in the combined state except in volcanic gases where it occurs in free state. It is the first element in the periodic table. It is the lightest of all the elements. It is the only element without neutrons i.e. Protium.
HYDROGEN AND ITS COMPOUNDS Introduction: It is the element with confusing position in the periodic table, as it resembles both I-A and VII-A group elements. Hydrogen should belong to the s-block as its electron configuration is 1s1 . The properties in which hydrogen resembles alkali metals are a) Like alkali metals it has one electron in s-orbital of valence shell. b) Like Alkali metals, it forms H+ ion. c) Like Alkali metals it forms monoxides and peroxides (H2O and H2O2). d) Like Alkali metals, it acts as reductant ) a reducing agent(.
HYDROGEN AND ITS COMPOUNDS Introduction: The properties in which Hydrogen resembles Halogens are a) Like Halogens it exists as diatomic molecule (H2) b) Like Halogens it gains one electron to attain inert gas configuration c) The I.E value of "H" is similar to that of Halogens. d) Like halogens it forms univalent anion i.e H .
HYDROGEN AND ITS COMPOUNDS Introduction: The ionization energy of H H bond is very high (1312 kJmol 1). This indicates that hydrogen has a low tendency to form H+ ions. Its ionization energy value is comparable to that of halogens. Hence, it forms diatomic molecules (H2), hydrides with elements, and a large number of covalent bonds. Since ionization energy is very high, hydrogen does not possess metallic characteristics (lustre, ductility, etc.) like metals.
HYDROGEN AND ITS COMPOUNDS Hydrogen has 3 isotopes. They are Theoretically the number of possible Hydrogen molecules is six. They are H2, D2, T2, HD, HT and DT. Ordinary Hydrogen contains only 0.02% Deuterium. Hydrogen and Deuterium can be separated by gas diffusion principle. Hydrogen is more reactive than Deuterium.
HYDROGEN AND ITS COMPOUNDS All the isotopes of Hydrogen have: a) same chemical properties; b) same atomic radii and c) same bond lengths. The isotopes of Hydrogen differ widely due to large difference in mass numbers. a) Number of neutrons ; b) Chemical reactivity; c) Physical properties; d) Nuclear radii and e) Bond energy. The properties or physical constants which are lower for hydrogen than that of deuterium are; i) Molecular weight; ii) Boiling point; iii) Melting point; iii) Bond energy and iv) Activation energy.
HYDROGEN AND ITS COMPOUNDS Reactivity What does it mean to be reactive? Elements that are reactive bond easily with other elements to make compounds. Some elements are only found in nature bonded with other elements. What makes an element reactive? o An incomplete valence electron level. All atoms (except hydrogen) want to have 8 electrons in their very outermost energy level (This is called the rule of octet.) Atoms bond until this level is complete. Atoms with few valence electrons lose them during bonding. Atoms with 5, 6, or 7 valence electrons gain electrons during bonding.
HYDROGEN AND ITS COMPOUNDS Reactivity Chemical reactivity is higher for hydrogen than that of Deuterium. On electrolysis of water, H2 is released six times faster than that of D2 . The radioactive isotope of Hydrogen is Tritium. As the mass number increases the chemical reactivity decreases. So chemically least reactive isotope of Hydrogen is Tritium. o It is a beta emitter and its half life is 12.26 years o 1T3 2 He3 + - 1e 0( - particle) Tritium is harmless, as it emits only low energetic -radiation and will not emit harmful -rays. In the study of mechanism of various chemical reactions, Deuterium and Tritium are used as tracers.
HYDROGEN AND ITS COMPOUNDS Production of hydrogen Lab scale - zinc and dil HCl - Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) Industrial large scale Reaction of steam on hydrocarbons or coke at high temperatures in the presence of catalyst yields hydrogen. CnH2n+2(g) + n H2O(g) --(Ni/1270 K)--> n CO(g) + (3n+1) H2(g) e.g. CH4(g) + H2O(g) --(Ni/800oC)--> CO(g) + 3H2(g) The mixture of CO and H2 is called water gas.
HYDROGEN AND ITS COMPOUNDS Uses of Hydrogen Hydrogen is used i) in the manufacture of chemicals ii) in metallurgy Hydrogen is used as a reducing agent in the extraction of heavier metals like Molybdenum and Tungsten. WO3 + 3H2 W + 3H2O iii) as a source of atomic energy iv) as a fuel. The heat of combustion of H2 gas is high (242 KJ/mole). Hence it is used as an industrial fuel.
HYDROGEN AND ITS COMPOUNDS Uses of Hydrogen a) Synthesis of Ammonia by Haber's process. 2NH3(g) N2(g) + 3H2(g) b) Preparation of HCl Water H2(g) + Cl2(g) 2HCl(g) 2HCl(aq) c) Synthesis of Methyl alcohol d) In the production of Margarine.
HYDROGEN AND ITS COMPOUNDS Hydrides The binary compounds of hydrogen with other elements are called hydrides. The hydrides have the formula MHx or (MmHn) These are formed by; i) all main group elements except the noble gases. ii) All lanthanides and Actinides. iii) Transition elements such as Sc, Y, La, Ac, Tc, Zr, Hf and to lesser extent V, Nb, Ta, Cr, Cu and Zn
HYDROGEN AND ITS COMPOUNDS Hydrides On the basis of their physical and chemical properties and types of bonding hydride are classified into the following four types: 1) Ionic hydrides (or) saline hydrides. 2) Covalent (or) molecular hydrides. 3) Metallic (or) Interstitial hydrides. 4) Polymeric hydrides.
HYDROGEN AND ITS COMPOUNDS Ionic hydrides
HYDROGEN AND ITS COMPOUNDS Ionic hydrides These are formed by elements of group IA, IIA (except Be and Mg) and lanthanides by heating the metal in hydrogen. 573 K 2Na + H2 2NaH 673 K 2K + H2 2KH Ca + H2 CaH2 Sr + H2 SrH2 These are white colourless solids (crystalline) having high melting point and 1073 K 1173 K boiling point easily decomposed by water, alcohol, CO2 (or) SO2 Except LiH. all ionic hydrides decompose at 400 C-500 C (on strong heating) into their constituent elements.
HYDROGEN AND ITS COMPOUNDS Ionic hydrides Electrolysis of these molten hydrides produce hydrogen gas at anode. 2H-(molten) H2(g) + 2e This reaction provides chemical evidence for the presence of H in these hydrides This type of hydrides are formed only by elements with electronegativity value that is less than 2.1. Hydrogen compounds of high electropositive metals may be regarded as the metal hydride. The density of these hydrides is greater than that of the metal from which they are formed. These ionic hydrides are stoichiometric and have high heats of formation.
HYDROGEN AND ITS COMPOUNDS Ionic hydrides Thermal stability of ionic hydride decreases with increasing size of cations LiH NaH KH RbH CsH CaH2 SrH2 BaH2 USES OF IONIC HYDRIDES : Metal hydrides are used in laboratory to remove traces of water from solvents and inert gases such as N2 and Ar. Alkali metal hydrides are used to prepare other hydride compounds Like - LiAlH4, NaBH4 etc. Since they evolve hydrogen when heated and therefore ignite spontaneously they are used as solid fuels.
HYDROGEN AND ITS COMPOUNDS Covalent hydrides
HYDROGEN AND ITS COMPOUNDS COVALENT OR MOLECULAR HYDRIDES : Molecular hydrides are soft, have low melting point and low boiling point. Molecular hydrides are volatile in nature have low electrical conductivity. These hydrides are formed by the combination of elements of higher electronegatively as p-block elements. (IIIA to VIIA groups) Example NH3, HCl, CH4, H2O, HF, B2H6, AsH3 Their molecular formula can be written as MHn (or) MH8 n , n = group number of element in short form of periodic table.
HYDROGEN AND ITS COMPOUNDS COVALENT OR MOLECULAR HYDRIDES : The molecular hydrides are classified into three categories. o Electron precise compounds: In these compounds all the electrons of the atom are involved in bond formation. Example CH4 (methane), C2H6 (ethane) o Electron deficient compounds: In these compounds the available number of valence electrons are less than the number required for covalent bond formation. Example B2H6
HYDROGEN AND ITS COMPOUNDS COVALENT OR MOLECULAR HYDRIDES : oElectron - rich compounds: In these compounds the valence electrons on the central atom are more than that are required for bond formation. i.e. lone pair present on the central atom. Example NH3, H2O The bonds present in this hydrides are mostly covalent in character. Covalent hydrides like HF, HCl are partly ionic in nature. The molecules are held together by weak van der Waal s forces and hence have low melting point and boiling point.
HYDROGEN AND ITS COMPOUNDS Metallic hydrides
HYDROGEN AND ITS COMPOUNDS METALLIC HYDRIDES: These are interstitial compounds as the hydrogen atom occupy interstitial position in the metal lattices. This is due to small size of hydrogen atoms compared to metal atom. The finely divided metals obtained by thermal decomposition of these hydrides are used as catalyst and in metallurgy.
HYDROGEN AND ITS COMPOUNDS METALLIC HYDRIDES: Most of the d-block elements and f block elements form metallic hydrides on reacting with hydrogen. Example CrH, CrH2, ZnH2 Be and Mg of s-block and aluminium also form metallic hydrides. Metallic hydrides are normally prepared by heating the metal with hydrogen under high pressure. Most of the metallic hydrides are metallic conductors and have variable composition. These hydrides have properties similar to those of parent metals.
HYDROGEN AND ITS COMPOUNDS METALLIC HYDRIDES: Metallic hydrides are hard, have metallic lustre and have magnetic properties. The density of these hydrides is less than that of the parent elements. Metallic hydrides exhibit metallic properties and are powerful reducing agents. Metallic hydrides are non-stoichiometric compounds and their composition varies with temperature and pressure. In these hydrogen exist in the atomic rather than ionic form.
HYDROGEN AND ITS COMPOUNDS POLYMERIC HYDRIDES : These are solids containing molecules linked together in two (or) three dimensions by hydrogen bridge bonds. (BeH2)n, (MgH2)n and (AlH3)n
HYDROGEN AND ITS COMPOUNDS HYDROGEN PEROXIDE H2O2 is also known as oxygenated water. [O-O]2-bond is known as Peroxy bond. H2O H2O2 is a weak dibasic acid. Its molecular weight is 34 and its equivalent weight is 17. H2O2 Substances containing peroxy linkage are H2O2, Na2O2, BaO2, NaHO2, H2SO5, HNO4, H3PO5 Perchloric acid (HClO4), MnO2, PbO2 etc are not true peroxides as they do not contain peroxy bond. HClO4
HYDROGEN AND ITS COMPOUNDS The structure of H2O2 In the solid (crystalline) phase, H2O2 has an open book structure of H as shown: The H O O bond angle is 101o The dihedral angle is 90o The O - O bond length is 1.48 and the O - H is 0.97 . In liquid and solid states the bond lengths and bond angles are slightly changed due to hydrogen bonding. In crystalline H2O2 the dihedral angle is Structure and dimensions of H2O2in the: Solid phase (up) and b) gas phase (botton) reduced to 90 .
HYDROGEN AND ITS COMPOUNDS Uses Of H2O2 As bleaching agent for textiles, silk, wool, wood pulp etc. Mixture of H2O2 and hydrazine is used as rocket fuel. To restore color of old oil paints. As Oxidising agent Mixture of H2O2 and NH3 solution is used to bleach human hair into golden yellow color.
HYDROGEN AND ITS COMPOUNDS Reactivity On long standing or on heating H2O2 undergoes decomposition. The equation for the decomposition is 2 H2O2 2 H2O + O2 The substances which retard the decomposition of H2O2 are; Acetanilide, Glycerol, Alcohol, H3PO4 etc. o They act as negative catalysts. oThey are known as inhibitors (or) stabilisers. Alkalies, silica, MnO2, metals (Fe, Mn), graphite, alumina etc. catalyse the decomposition of H2O2. Dilute aqueous solution of H2O2 is fairly stable in acid Medium of alcohol or ester. Alkaline.
HYDROGEN AND ITS COMPOUNDS Precautions to be taken in storing H2O2. a) The glass bottle must have wax coating on the inner side, so that the surface is smooth. b) Plastic bottles are preferred to glass bottles. It is because the Na2O in glass being alkaline, catalyses the decomposition of H2O2. c) A small quantity of acetanilide or urea is added as a stabiliser.
HYDROGEN AND ITS COMPOUNDS Boron Hydrides (Hydrides of Boron) Boron can form many different types of hydrides; Borane (BH3), reacts violently with air and is easily oxidized. Borane occurs as a gaseous substance, It can forms Diborane (B2H6) by two BH3 molecules combined with each other. Borane has only six valence electrons so it is unstable compound (does not follow a complete octet rule). Borane is formed via thermal decomposition of diborane (B2H6 ). Diborane (B2H6) A gas (bp 92.6 C) that is flammable in air and instantly hydrolyzed by H2O: The oxidation of B2H6 is extremely exothermic.
HYDROGEN AND ITS COMPOUNDS Structure and Bonding in Boranes The structure and bonding in boranes is unlike those of other hydrides (e.g. CH4); There are not sufficient electrons to allow formation of conventional two- electron bonds > electron deficient bonds In the valence bond model the B H B bridge in diborane It can be described as a three-center-two-electron bond (3c-2e bond):
HYDROGEN AND ITS COMPOUNDS Neutral Boranes Beside diborane there is a large number of borane cluster compounds:
HYDROGEN AND ITS COMPOUNDS Structure and Bonding in Boranes To account for the structure and bonding in higher borane there are a total of five structurally different bonding elements present: