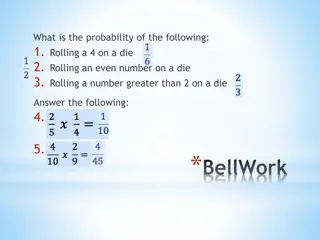Understanding Adverse Events Following Immunization (AEFI)
Adverse Events Following Immunization (AEFI) are medical incidents that occur after immunization, potentially caused by the vaccine, leading to unfavorable symptoms. Pharmacovigilance plays a crucial role in detecting, assessing, and preventing these events. AEFI can impact immunization programs at
4 views • 40 slides
Overview of UW Clinical Trial Office Budget Review
UW Clinical Trial Office conducts budget reviews to ensure compliance and financial accountability in clinical trials. The office collaborates with various departments to manage billing compliance, financial risks, and institutional policies. The primary focus is on avoiding patient billing errors,
0 views • 17 slides
India Alliance Clinical & Public Health fellowship in India
India Alliance Clinical & Public Health fellowship in India\n\nIndia Alliance Clinical and Public Health Research Fellowships are for Health researchers with an MD, MS, MPH, or an equivalent clinical or public health degree, who can apply for the DBT\/Wellcome Trust India Alliance Clinical and Publi
0 views • 5 slides
Clinical Escalation: Building Effective Communication in Maternity Units
Exploring the importance of clinical escalation in maternity units, this session outlines the components and practices involved in identifying, communicating, and acting upon clinical concerns. It emphasizes recognizing deviation from normality, effective communication, and taking appropriate action
0 views • 27 slides
Training Models in Clinical Psychology: A Comprehensive Overview
Clinical psychology training models such as the Scientist-Practitioner Model have evolved over the years to integrate science and practical expertise. The Scientist-Practitioner Model, originating in 1949, emphasizes the fusion of scientific knowledge and clinical application. It faced criticism for
1 views • 98 slides
Ethical Issues in Clinical Pharmacy Research by Dr. Haider Raheem Mohammad
Research ethics play a crucial role in clinical trials and therapeutic research in the field of pharmacy. From discovery to validation, all medicines undergo rigorous evaluation processes to ensure safety, efficacy, and freedom from adverse effects. Clinical trials in both animals and humans are ess
0 views • 20 slides
Understanding Probability in Events
Explore concepts of probability in various events like rolling a die, compound events, simple events, and spinner probability. Learn how to calculate probabilities of different outcomes and understand the difference between single and compound events. Discover key principles in probability theory an
0 views • 13 slides
Advanced Clinical Practice Framework and Pillars of Practice
The document discusses the advanced clinical practice framework and the four pillars of practice which include leadership & management, clinical practice, education, and research. It emphasizes the importance of core capabilities and area-specific competence in advanced clinical practice. The role o
2 views • 8 slides
Digital Health Technology-Derived Clinical Outcome Assessments in Regulatory Decision-Making
This session discusses the landscape of DHT-derived novel endpoints in clinical research, focusing on considerations for regulatory decision-making. It explains the use of digital health technology in clinical outcome assessments and highlights the potential benefits of digitally collected COAs, suc
0 views • 42 slides
Objective Structured Clinical Examination (OSCE): A Modern Approach to Assessing Clinical Competence
The Objective Structured Clinical Examination (OSCE) is a modern examination method widely used in the field of health science to evaluate clinical skill performance. It involves stations where medical students interact with simulated patients to demonstrate competencies such as history taking, phys
1 views • 40 slides
NIMH Clinical Research Education and Monitoring Program Overview
NIMH's Clinical Monitoring and Clinical Research Education, Support, and Training Program (CREST) aims to ensure the proper conduct, recording, and reporting of clinical trials. This program includes clinical monitoring plans, guidelines for site monitoring activities, and independent clinical monit
1 views • 29 slides
Understanding Statistical Methods for Clinical Endpoints in Diabetes Research
This educational slide module delves into fundamental statistics for analyzing clinical endpoints in diabetes research. It covers the choice of statistical methods, the distinction between statistical and clinical significance, and the importance of different endpoints in evaluating clinical benefit
1 views • 37 slides
Understanding Ethics in Clinical Trials: A Comprehensive Overview
Explore the historical context, important ethical guidelines, and the ethical framework with 7 principles in the field of clinical trials. Learn about key trials, ethical considerations, and guidelines governing human subject research in clinical medicine. Delve into the critical aspects such as inf
1 views • 15 slides
Understanding Non-Clinical Development in Therapeutic Innovation
The European Patients Academy on Therapeutic Innovation focuses on the non-clinical development phase of medicine, delving into efficacy assessment, safety evaluation, and manufacturing process considerations. Non-clinical studies are essential for decision-making in clinical trials, marketing appli
1 views • 26 slides
UVA Clinical Nursing Preceptor Training Regulations
Virginia Board of Nursing regulations outline the role of clinical preceptors in supervising nursing students, ensuring safe client care, and enhancing clinical learning experiences. Preceptors serve as teachers, mentors, and role models, linking classroom knowledge with practical skills. Faculty me
0 views • 36 slides
Importance of Communication and Counseling Skills in Clinical Settings
Dr. Leena Baghdadi, an Assistant Professor and Clinical Epidemiologist, emphasizes the significance of effective communication and counseling skills in clinical practice. The content discusses the objectives of understanding these skills, barriers, and practical examples of counseling. Highlighted b
0 views • 27 slides
BMC Clinical Trials Office Goals and Initiatives
The BMC Clinical Trials Office (CTO) is dedicated to supporting clinical and human research at Boston Medical Center by providing leadership and expertise in research, finance, and administration. The CTO reviews, negotiates, and approves protocols, supports grants and contracts, ensures accurate cl
0 views • 17 slides
Understanding Clinical Trials: Types and Designs
Clinical trials are essential research studies that evaluate new tests and treatments to improve human health outcomes. They involve various phases, designs, and purposes, such as treatment trials, prevention trials, and observational studies. Different types of clinical trial designs include experi
7 views • 18 slides
Veterinary Medicinal Products Clinical Trials in Ireland
This presentation covers the process and regulations surrounding clinical trials and testing of veterinary medicinal products in Ireland. Topics include the application process, HPRA evaluation, fees, scope of legislation, and the purpose of clinical trials. Feedback from a 2016 HPRA survey on clini
3 views • 17 slides
Understanding Clinical Validation and DRG Validation in Healthcare
Clinical validation ensures that diagnoses documented in a patient's record align with accepted clinical criteria, while DRG validation focuses on matching hospital-coded information with physician descriptions and patient records. Clinical validation involves a review by clinicians to confirm the p
1 views • 39 slides
Dr. Chris Webb's December 2020 Clinical Supervisors Report
A structured Clinical Supervisors Report (CSR) for Dr. Chris Webb in December 2020. This report provides insight into the clinical supervision process within various posts and includes valuable information from the Clinical Supervisor.
0 views • 26 slides
Understanding Evidence-Based Medicine and Clinical Decision-Making
European Patients Academy on Therapeutic Innovation emphasizes the importance of Evidence-Based Medicine (EBM) in providing optimum clinical care. EBM involves systematic review and utilization of clinical research for informed decision-making, benefiting patients in disease management and treatment
7 views • 20 slides
Navigating Challenges in Clinical Rotations for Health Professions Faculty
This presentation highlights the challenges and choices faced by health professions faculty in clinical rotations, focusing on identifying opportunities and resources for limited clinical experiences. The content covers fundamental terminology, options available, and strategies such as on-site and r
1 views • 18 slides
Guide to Participating in STAR Events in Georgia FCCLA
Discover the significance of STAR Events in Georgia FCCLA, competitive events acknowledging proficiency in various projects, leadership, and career readiness. Learn how to select individual or team events, choose a suitable project, and initiate your project effectively. Empower yourself to excel in
0 views • 19 slides
Essential Aspects of the Clinical Interview in Psychology
Clinical interviews play a crucial role in the assessment conducted by clinical psychologists, showcasing essential qualities like validity, reliability, and clinical utility. Understanding the importance of feedback and honing general and specific skills as an interviewer are key components in cond
1 views • 17 slides
Comprehensive Guidance for Clinical Staff on Complaints Handling Process
Developed for clinical professionals, this guidance provides support for participating in complaints investigations and writing clinical reports. It aims to clarify the definition of clinical judgment and outlines the key elements of handling complaints effectively. The module encourages collaborati
0 views • 19 slides
Clinical Trial Comparison of Immediate vs Deferred Antiretroviral Therapy in HIV-Infected Individuals
This study compared immediate initiation of antiretroviral therapy (ART) with deferred treatment in HIV-infected individuals with CD4+ count >500 cells/mm3. Results showed a significant reduction in primary composite endpoint events in the immediate ART group compared to the deferred ART group. Seri
0 views • 4 slides
Clinical Research Guidelines and Regulations Overview
Clinical research encompasses various guidelines and regulations to ensure the protection of human subjects and the credibility of study results. Key aspects include Good Clinical Practice (GCP) standards, Title 45 of the Code of Federal Regulations (CFR) Part 46, and additional CFR sections for cli
0 views • 46 slides
Ohio Clinical Alliance: Transforming Clinical Experiences
The Ohio Clinical Alliance, through collaborative partnerships, aims to enhance clinical preparation for educators. The leadership team comprises various representatives and organizations committed to improving student learning. Their activities include retreats and meetings to ensure effective comm
0 views • 27 slides
RMS PTO Board Meeting Update and Event Schedule
The recent RMS PTO board meeting covered various topics such as budget tracking, upcoming events, and recent activities. Discussions included PayPal usage, budget allocations for future fundraisers, expenses, and a review of recent events. The meeting also outlined an array of events planned for the
0 views • 7 slides
Understanding Probability of Compound Events
Compound events involve multiple outcomes occurring together, such as independent events where one outcome does not affect another, and dependent events where one outcome affects the other. Complement sets, card probabilities, and mutually exclusive events are also discussed, providing a comprehensi
0 views • 15 slides
Safety Practices and Reporting in Clinical Research
Safety practices and reporting in clinical research are crucial for ensuring the rights, safety, and well-being of trial subjects. This includes monitoring safety, reporting adverse events promptly, and following regulatory requirements. Investigators play a vital role in assuring subject safety and
0 views • 33 slides
Perception and Awareness of Clinical Research in Trial Participants and the Public of Andhra Pradesh
This study focuses on understanding the perception and awareness of clinical research among trial participants and the general public in Andhra Pradesh. It highlights the importance of creating awareness about clinical research, previous study results, public attitudes towards clinical trials, and e
0 views • 24 slides
Comparison of Professional Behaviors in Clinical Education
Professional behavior characteristics play a crucial role in enhancing student learning during clinical education. This study examines the differences in reported importance and frequency of professional behaviors between credentialed and non-credentialed clinical instructors. The background outline
0 views • 28 slides
Noise Control Measures and Community Preferences at the Corn Exchange
Various images and references discuss the history of amplified music events at the Corn Exchange, noise complaints leading to control methods by the FTC, impacts on events and community preferences post-control, and survey findings on participant demographics, noise effects, and desired public event
0 views • 19 slides
Understanding Adverse Events and Unanticipated Problems in Clinical Research
Explore the significance of adverse event reporting in clinical research, including definitions of adverse events (AEs) and serious adverse events (SAEs), as well as unanticipated problems (UAPs). Learn about the responsibilities of Investigators, Clinical Research Coordinators (CRCs), and Sponsors
0 views • 63 slides
Enhancing Clinical Academic Collaboration Between Universities and NHS Trusts
Clinical academics play a crucial role in integrating clinical practice, research, and education within the NHS. Collaboration between universities and NHS trusts is key to ensure clinical academics address the right questions for patient care and societal benefit. Challenges include an aging clinic
0 views • 29 slides
Brain Oxygen Optimization in Severe TBI: Adverse Events Analysis
This study led by Dr. Robert Silbergleit focuses on adverse events in brain oxygen optimization for severe Traumatic Brain Injury (TBI) patients. It outlines key points for reporting adverse events, discusses relatedness algorithms, and presents scenarios to analyze adverse events occurrence post-en
0 views • 24 slides
Understanding Clinical Trials: Phases, Types, and Definitions
Clinical trials play a crucial role in advancing medical research and treatment options. This comprehensive guide covers the basics of clinical trials, including their definition, phases, types, and key definitions like IND, IDE, NDA, and more. Discover how different phases of trials work, the vario
0 views • 16 slides
Official Kayaking Rules for Michigan Events
Official rules for kayaking events in Michigan including information on events offered, uniforms, equipment requirements, and general rules for athletes. Athletes can enter up to three events but must choose between Traditional or Unified events. The rules cover attire, required equipment such as li
0 views • 14 slides