Understanding Primary Bonding in Solids: Importance and Types
Exploring the fundamental concept of bonding in solids, this lecture delves into primary bonding, which includes covalent, ionic, and metallic bonds. The discussion highlights how different types of bonds impact the properties of materials such as metals, ceramics, and polymers. By understanding the nature of bonding, we can decipher why materials exhibit specific characteristics and behaviors.
Download Presentation

Please find below an Image/Link to download the presentation.
The content on the website is provided AS IS for your information and personal use only. It may not be sold, licensed, or shared on other websites without obtaining consent from the author. Download presentation by click this link. If you encounter any issues during the download, it is possible that the publisher has removed the file from their server.
E N D
Presentation Transcript
Lecture 2 Bonding in solids Primary Bonding 1
Recap We discussed the structure of materials from the materials tetrahedron point of view because the structure is integrated intricately related to properties, processes, and applications. So, earlier, we classified materials in four categories, the first category is metals and alloys, second was ceramics and glasses, the third category was polymers and elastomers, and the fourth category was hybrids or composites. 2
Metals are strong, ductile, and tough. However, they have poor corrosion resistance, they have high thermal conductivity. Ceramics, on the other hand, are brittle, but they are very strong. However, they have low electrical and thermal conductivity. Polymers, on the other hand, are soft, light, they can be stretched too long distances.They are also tough, and they are also very corrosion resistant, but they are not very good for high-temperature applications. Composites, on the other hand, are manufactured by mixing two contrasting materials to leverage the advantage of both the different classes of materials. So far, we discussed one way of classifying the materials, and now we are going to discuss the material classification based on atomic bonding. 3
Bonding A chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds. The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bonds. The strength of chemical bonds varies considerably; there are "strong bonds" or "primary bonds" such as covalent, ionic and metallic bonds." or "secondary bonds" such as dipole dipole interactions, the London dispersion force and hydrogen bonding. 4
Bonding For example, metals and alloys are bonded via metallic bonds. Moreover, it is the nature of these bonds, which is very crucial in imparting the properties which these materials have. Metals have high electrical conductivity, high thermal conductivity, malleability, or ductility because of metallic bonding. Ionic bonds or partially covalent bonds bond ceramics and glasses. For example, sodium chloride would be highly ionic bonding; bonding in silicon carbide and zinc oxide will have partially ionic and covalent in bonding. Ceramics are strong, and they have low electrical, thermal conductivity, and they have a low coefficient of thermal expansion because ionic bonds or covalent bonds bond them. Polymers, on the other hand, they have a mixture of covalent and secondary bonding and the secondary bonding plays an important role in determining the properties. 5
Bonding Each and everything in this universe tries to become stable. This happens only by losing energy. In an interaction between two types of matter, one exerts a force on another. The energies between the two types of matter depend on the nature of the force between them. If the force is attractive, the energy decreases. On the other hand, if it s repulsive in nature then the energy increases. In cases where the force is attractive, the two atoms get bound together. Such a force is what we call a chemical bond. Thus, we can define chemical bonding as follows: The attractive force which holds various constituents (atom, ions, etc.) together and stabilizes them by the overall loss of energy is chemical bonding. 6
Primary and Secondary bonds The atoms and molecules in a solid state are more closely packed as compared to gaseous and liquid states and are held together by strong mutual forces of attraction. These interatomic forces are electrostatic in nature and depend upon the electronic structure of matter. When atoms come closer and finally unite to form molecules their electrons rearrange themselves in such a way as to achieve a stable configuration. This arrangement of electrons gives rise to different types of bonds due to which atoms are held together. Bonds can be broadly classified as: 1. Primary bonds 2. Secondary bonds 7
Primary and Secondary bonds Primary Bonds Primary bonds are formed when the bonding process involves a transfer or sharing of electrons between atoms to form a more stable electron configuration. Primary bonds are Strong in comparison to secondary bonds. Secondary bonds Secondary bonds are formed from the subtle attraction forces between positive and negative charges. Secondary bonds are weak in comparison to primary bonds. Primary bonding occurs when electrons are lost or gained so that the outer shell is filled. This occurs through two mechanisms - Ionic , Covalent and Metallic bonding. Secondary bonding occurs due to forces produced by atomic or molecular dipoles. These are broadly classified as Van der Waal's forces ,hydrogen bonds and the London dispersion force 8
Ionic Bonding Ionic Bonding : Ionic bonding involves the transfer of electrons between atoms. In such cases, one atom loses an electron and the other atom gains an electron. Or Positive and negative ions form when a metal reacts with a non-metal, by transferring electrons. The oppositely charged ions are strongly attracted to each other, forming ionic bonds. When an electron transfer occurs, one atom has a negative charge making it the anion. On the other hand, the other atom has a positive charge making it the cation. For instance, NaCl or sodium chloride is a compound formed from ionic bonds between positively charged Na+ ions and negatively charged Cl- ions. 9
Ionic Bond This compound is hard but brittle and does not conduct electricity when it is solid but does so when in mixed in a solution or in liquid state. Furthermore, it has a very high melting point, in other words, strong heat is required to break the bonds between the constituent ions. All these strong characteristics of this compound are attributed to it by the presence of strong ionic bonds between its constituent elements. The ionic bond is the electrostatic force of attraction between two oppositely charged ions. Ionic bonds join metals to nonmetals. Ionic bonding is non-directional because an ion has the same attraction from all directions for an ion of opposite charge. Explanation: ... This means that packing efficiency in the crystal lattice determines the arrangement of ions in a crystal. There is no preferred direction for the bonds. Examples : Sodium chloride (NaCl), Sulfuric Acid (H2SO4) 10
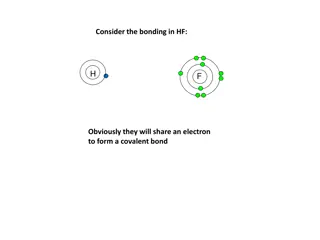
Covalent Bond Covalent bonds are those bonds formed when electrons are shared between elements giving rise to compounds. The covalent bond is also called a shared bond. These bonds join nonmetals to nonmetals. These bonds enable the constituent elements to complete their incomplete noble gas configuration. For example, the dioxygen molecule is formed from covalent bonds between two oxygen atoms. Each oxygen atom is two electrons short of the next noble gas configuration, which is of the neon atom. Therefore when these atoms come closer and share two electrons each, they give rise to a double covalent bond between the two shared electron pairs of the atoms. 11
Covalent Bond Covalent bonds are also possible for single and triple bonds where bonds are formed between one and three pairs of electrons respectively. These bonds are directional and generally insoluble in water. Diamond, the hardest known naturally occurring substance on Earth, is formed from covalent bonds between carbon atoms arranged in a 3D structure. A covalent bond is formed by the overlap of half- filled atomic orbitals which have definite directions. Hence covalent bond is directional. In ionic compounds, each ion is surrounded by a number of oppositely charged ions and hence there is no definite direction. Examples : Methane (CH4), Hydrochloric acid (HCl) 12
Metallic Bond Metallic bonds, as the name suggests, are bonds found only in metals.Metallic bonds are the chemical bonds that join metals to metals. Metals are elements of the electropositive nature, thus it is very easy for the constituent atoms to lose their outer shell electrons and form ions. Metallic bond is a term used to describe the collective sharing of a sea of valence electrons between several positively charged metal ions. Metallic bonding is a type of chemical bonding and is responsible for several characteristic properties of metals such as their shiny lustre, their malleability, and their conductivities for heat and electricity. Both metallic and covalent bonding can be observed in some metal samples. For example, covalently bonded gallium atoms tend to form crystal structures that are held together via metallic bonds. 13
Metallic Bond The factors that affect the strength of a metallic bond include: Total number of delocalized electrons. Magnitude of positive charge held by the metal cation. Ionic radius of the cation An illustration describing the way electrons are delocalized over a rigid lattice of metal ions in a metallic bond is provided below. Metallic Bonds involve a Sea of Electrons that are Delocalized over a Rigid Lattice of Positively Charged Metal Ions 14
Metallic Bond Example Metallic Bonding in Sodium The electron configuration of sodium is 1s22s22p63s1; it contains one electron in its valence shell. In the solid-state, metallic sodium features an array of Na+ ions that are surrounded by a sea of 3s electrons. However, it would be incorrect to think of metallic sodium as an ion since the sea of electrons is shared by all the sodium cations, quenching the positive charge. 15
Metallic Bond The softness and low melting point of sodium can be explained by the relatively low number of electrons in the electron sea and the relatively small charge on the sodium cation. For example, metallic magnesium consists of an array of Mg2+ ions. The electron sea here contains twice the number of electrons than the one in sodium (since two 3s electrons are delocalized into the sea). Due to the greater magnitude of charge and the greater electron density in the sea, the melting point of magnesium (~650oC) is significantly higher than that of sodium. Metallic bonds are not broken when the metal is heated into the melt state. Instead, these bonds are weakened, causing the ordered array of metal ions to lose their definite, rigid structure and become liquid. However, these bonds are completely broken when the metal is heated to its boiling point. Examples : Iron, cobalt, calcium and magnesium, silver, gold, barium, platinum, chromium, copper, zinc, sodium etc. 16
Metallic Bond The properties of metals that are a consequence of metallic bonding include: Malleability Ductility High melting and boiling point High electrical and thermal conductivity Metallic lustre 17
Difference among Ionic bond, Covalent bond, and Metallic bond IONIC BOND COVALENT BOND METALLIC BOND The attraction of metal cations/atoms and delocalized electrons Binding energy is less than covalent and ionic bond Has high electrical conductivity Non-directional bond Present only in one state: solid-state Mouldable High melting point Ductile High boiling point Occurs during the transfer of electrons Occurs when 2 atoms share their valence electrons Binding energy is higher than the metallic bond Binding energy is higher than the metallic bond Low conductivity Very low conductivity Non-directional bond Present only in one state: solid-state Unmoldable Higher melting point Non-ductile Higher boiling point Directional bond Present only in all 3 states: solid, liquid, gases Unmoldable Lower melting point Non-ductile Lower boiling point 18
Summary Bonding is determined by how electrons (Valence ) are handled by atoms when they are ensembled together. Primary bonding was discussed. Differences among Ionic, covalent and metallic Bonding were discussed. 19