National Taskforce on NCHD Workforce - Improving the Training Environment
The National Taskforce on NCHD Workforce aims to enhance the training experience, well-being, and work-life balance for Non-Consultant Hospital Doctors (NCHDs) in Ireland. The taskforce focuses on four key areas: addressing on-site working structures, fostering a culture of education and training, o
0 views • 19 slides
Overview of UW Clinical Trial Office Budget Review
UW Clinical Trial Office conducts budget reviews to ensure compliance and financial accountability in clinical trials. The office collaborates with various departments to manage billing compliance, financial risks, and institutional policies. The primary focus is on avoiding patient billing errors,
0 views • 17 slides
Dog trainer Vero Beach
All Training packages require a NON REFUNDABLE Deposit of $500.00 upon booking. Even in the event your dog becomes sick injured or passes away before training. We only reserve a certain amount of training spots each month. We will however honor your deposit or put it towards boarding credit for the
0 views • 6 slides
India Alliance Clinical & Public Health fellowship in India
India Alliance Clinical & Public Health fellowship in India\n\nIndia Alliance Clinical and Public Health Research Fellowships are for Health researchers with an MD, MS, MPH, or an equivalent clinical or public health degree, who can apply for the DBT\/Wellcome Trust India Alliance Clinical and Publi
0 views • 5 slides
Clinical Escalation: Building Effective Communication in Maternity Units
Exploring the importance of clinical escalation in maternity units, this session outlines the components and practices involved in identifying, communicating, and acting upon clinical concerns. It emphasizes recognizing deviation from normality, effective communication, and taking appropriate action
0 views • 27 slides
Training Models in Clinical Psychology: A Comprehensive Overview
Clinical psychology training models such as the Scientist-Practitioner Model have evolved over the years to integrate science and practical expertise. The Scientist-Practitioner Model, originating in 1949, emphasizes the fusion of scientific knowledge and clinical application. It faced criticism for
1 views • 98 slides
Ethical Issues in Clinical Pharmacy Research by Dr. Haider Raheem Mohammad
Research ethics play a crucial role in clinical trials and therapeutic research in the field of pharmacy. From discovery to validation, all medicines undergo rigorous evaluation processes to ensure safety, efficacy, and freedom from adverse effects. Clinical trials in both animals and humans are ess
0 views • 20 slides
Understanding Postdoctoral Training in Psychology
Postdoctoral training in psychology plays a crucial role in enhancing career prospects and skills development for graduate students. This training, typically lasting 1-2 years, offers specialized clinical or research training beyond the doctoral degree, increasing marketability and potential for boa
0 views • 28 slides
Advanced Clinical Practice Framework and Pillars of Practice
The document discusses the advanced clinical practice framework and the four pillars of practice which include leadership & management, clinical practice, education, and research. It emphasizes the importance of core capabilities and area-specific competence in advanced clinical practice. The role o
2 views • 8 slides
Digital Health Technology-Derived Clinical Outcome Assessments in Regulatory Decision-Making
This session discusses the landscape of DHT-derived novel endpoints in clinical research, focusing on considerations for regulatory decision-making. It explains the use of digital health technology in clinical outcome assessments and highlights the potential benefits of digitally collected COAs, suc
0 views • 42 slides
Objective Structured Clinical Examination (OSCE): A Modern Approach to Assessing Clinical Competence
The Objective Structured Clinical Examination (OSCE) is a modern examination method widely used in the field of health science to evaluate clinical skill performance. It involves stations where medical students interact with simulated patients to demonstrate competencies such as history taking, phys
1 views • 40 slides
NIMH Clinical Research Education and Monitoring Program Overview
NIMH's Clinical Monitoring and Clinical Research Education, Support, and Training Program (CREST) aims to ensure the proper conduct, recording, and reporting of clinical trials. This program includes clinical monitoring plans, guidelines for site monitoring activities, and independent clinical monit
1 views • 29 slides
Understanding Non-Aqueous Solvents: Types and Classification
Inorganic non-aqueous solvents play a crucial role in chemical research and industry. This article by Dr. Princy K.G. delves into the classification of solvents based on protonicity, polarity, and aqueous vs. non-aqueous nature. It explores the types of non-aqueous solvents, such as protonic and non
1 views • 29 slides
Understanding Statistical Methods for Clinical Endpoints in Diabetes Research
This educational slide module delves into fundamental statistics for analyzing clinical endpoints in diabetes research. It covers the choice of statistical methods, the distinction between statistical and clinical significance, and the importance of different endpoints in evaluating clinical benefit
1 views • 37 slides
Understanding Ethics in Clinical Trials: A Comprehensive Overview
Explore the historical context, important ethical guidelines, and the ethical framework with 7 principles in the field of clinical trials. Learn about key trials, ethical considerations, and guidelines governing human subject research in clinical medicine. Delve into the critical aspects such as inf
1 views • 15 slides
Understanding Non-Clinical Development in Therapeutic Innovation
The European Patients Academy on Therapeutic Innovation focuses on the non-clinical development phase of medicine, delving into efficacy assessment, safety evaluation, and manufacturing process considerations. Non-clinical studies are essential for decision-making in clinical trials, marketing appli
1 views • 26 slides
UVA Clinical Nursing Preceptor Training Regulations
Virginia Board of Nursing regulations outline the role of clinical preceptors in supervising nursing students, ensuring safe client care, and enhancing clinical learning experiences. Preceptors serve as teachers, mentors, and role models, linking classroom knowledge with practical skills. Faculty me
0 views • 36 slides
BMC Clinical Trials Office Goals and Initiatives
The BMC Clinical Trials Office (CTO) is dedicated to supporting clinical and human research at Boston Medical Center by providing leadership and expertise in research, finance, and administration. The CTO reviews, negotiates, and approves protocols, supports grants and contracts, ensures accurate cl
0 views • 17 slides
Understanding Non-Firm Quantities in Electricity Markets
Non-Firm Quantities in electricity markets involve units with non-firm access not being compensated for their non-firm capacity not getting accommodated on the system. The concept of Firm Access Quantity plays a key role in determining compensation levels for units, with differences in implementatio
0 views • 6 slides
Understanding Non-Compete Agreements: Enforceability and Requirements
Non-compete agreements are commonly used in the United States to protect businesses from competition by former employees. To be enforceable, these agreements must meet certain requirements, including independent consideration, protection of legitimate business interests, and reasonableness in scope,
0 views • 26 slides
Understanding Evidence-Based Medicine and Clinical Decision-Making
European Patients Academy on Therapeutic Innovation emphasizes the importance of Evidence-Based Medicine (EBM) in providing optimum clinical care. EBM involves systematic review and utilization of clinical research for informed decision-making, benefiting patients in disease management and treatment
7 views • 20 slides
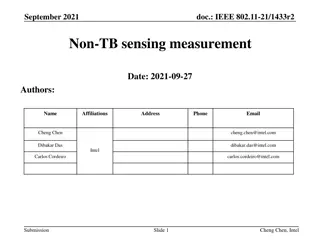
Comparison of Trigger-based vs. Non-Trigger-based Sensing Measurement in IEEE 802.11
The document discusses the differences between Trigger-based (TB) and Non-Trigger-based (Non-TB) sensing measurement instances in IEEE 802.11 standards, focusing on who initiates the sensing measurement. TB sensing is initiated by the AP, while Non-TB sensing is initiated by a non-AP STA, enabling o
6 views • 13 slides
General Clinical Research Unit (GCRU) Services Overview
The General Clinical Research Unit (GCRU) under Ridiane Denis offers support for a wide range of active protocols in various research areas like endocrinology, Alzheimer's, oncology, and more. GCRU provides clinical and laboratory services including sample processing, infusion, biopsy, urine analysi
0 views • 5 slides
Essential Aspects of the Clinical Interview in Psychology
Clinical interviews play a crucial role in the assessment conducted by clinical psychologists, showcasing essential qualities like validity, reliability, and clinical utility. Understanding the importance of feedback and honing general and specific skills as an interviewer are key components in cond
1 views • 17 slides
Greater Horizon Training Institute - Allied Health Programs
Greater Horizon Training Institute offers a range of certified preparation programs in Allied Health, including Nursing Assistant, Patient Care Technician, Clinical Medical Assistant, and Medical Billing/Coding Specialist. The institute strives to provide quality training implemented by dedicated fa
1 views • 8 slides
Enhancing Clinical Reasoning Training in Medical Education
Integrating research activities into educational programs is essential for advancing clinical reasoning training in medical education. This study explores different teaching approaches for clinical reasoning, focusing on addressing traditional teaching drawbacks and implementing simulation encounter
0 views • 31 slides
Enhancing Human Subjects Research Through NIH Policy Changes
The National Institutes of Health (NIH) is implementing reforms and initiatives to improve the stewardship of research involving human subjects, particularly in the context of clinical trials. These changes include new forms for data collection, training in Good Clinical Practice, use of a single In
0 views • 13 slides
Clinical Research Guidelines and Regulations Overview
Clinical research encompasses various guidelines and regulations to ensure the protection of human subjects and the credibility of study results. Key aspects include Good Clinical Practice (GCP) standards, Title 45 of the Code of Federal Regulations (CFR) Part 46, and additional CFR sections for cli
0 views • 46 slides
Ohio Clinical Alliance: Transforming Clinical Experiences
The Ohio Clinical Alliance, through collaborative partnerships, aims to enhance clinical preparation for educators. The leadership team comprises various representatives and organizations committed to improving student learning. Their activities include retreats and meetings to ensure effective comm
0 views • 27 slides
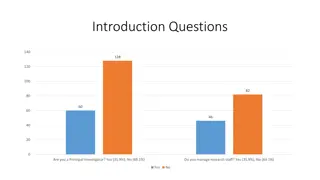
Survey Results on Research Training and Educational Support in Clinical Research Settings
This presentation showcases survey data on various aspects of research training and educational support in clinical research environments, including questions on being a Principal Investigator, managing research staff, training adequacy, study types, tenure at the University of Chicago, training pre
0 views • 35 slides
Effective Training Manual for Integrating People with Disabilities into the European Labour Market
This comprehensive training manual provides guidance for trainers on implementing the innovative JobCircuit model by PhoenixKM, Belgium. It covers didactic guidance, planning and preparing training sessions, facilitating engaging training environments, and evaluating training impact. The manual outl
0 views • 20 slides
Capacity Building and Training for Extension Workers and Farmers
Capacity building involves compulsory training and skills development for extension workers and farmers, along with effective reward systems. Training is essential for individuals to acquire new skills and knowledge, fitting them for their roles. The process includes developing competencies required
0 views • 18 slides
Perception and Awareness of Clinical Research in Trial Participants and the Public of Andhra Pradesh
This study focuses on understanding the perception and awareness of clinical research among trial participants and the general public in Andhra Pradesh. It highlights the importance of creating awareness about clinical research, previous study results, public attitudes towards clinical trials, and e
0 views • 24 slides
Understanding Clinical Psychology: Definition, Training, and Models
Clinical psychology, first introduced in 1907, encompasses a diverse field focusing on the study, assessment, and treatment of psychological issues. The APA Division 12 defines clinical psychology as integrating science, theory, and practice to understand and alleviate maladjustment. Training typica
0 views • 26 slides
Challenges and Solutions in Clinical Science Training
Challenges facing evidence-based mental health care provision in the US include incomplete knowledge, poor dissemination, and disparities in accessibility. Clinical science training aims to prepare graduates to engage in generating new scientific knowledge, providing clinical care, disseminating kno
0 views • 15 slides
Clinical Instructor Training and Nursing Education Guidelines
Clinical Instructor Training program and Nursing Education guidelines at Vanderbilt University Medical Center including training requirements, feedback procedures, conflict of interest policies, clinical reminders, bedside RN responsibilities, and patient assignment protocols.
0 views • 18 slides
Comparison of Professional Behaviors in Clinical Education
Professional behavior characteristics play a crucial role in enhancing student learning during clinical education. This study examines the differences in reported importance and frequency of professional behaviors between credentialed and non-credentialed clinical instructors. The background outline
0 views • 28 slides
Enhancing Clinical Academic Collaboration Between Universities and NHS Trusts
Clinical academics play a crucial role in integrating clinical practice, research, and education within the NHS. Collaboration between universities and NHS trusts is key to ensure clinical academics address the right questions for patient care and societal benefit. Challenges include an aging clinic
0 views • 29 slides
Non-Academic Requirements (NARs) for Clinical Placements
Non-Academic Requirements (NARs) are essential documents mandated for clinical placements to ensure the safety of students, staff, and patients. These requirements include criminal record checks, communicable disease forms, tuberculosis testing, vaccinations, and other health and safety training. Co
0 views • 18 slides
Understanding Clinical Trials: Phases, Types, and Definitions
Clinical trials play a crucial role in advancing medical research and treatment options. This comprehensive guide covers the basics of clinical trials, including their definition, phases, types, and key definitions like IND, IDE, NDA, and more. Discover how different phases of trials work, the vario
0 views • 16 slides